 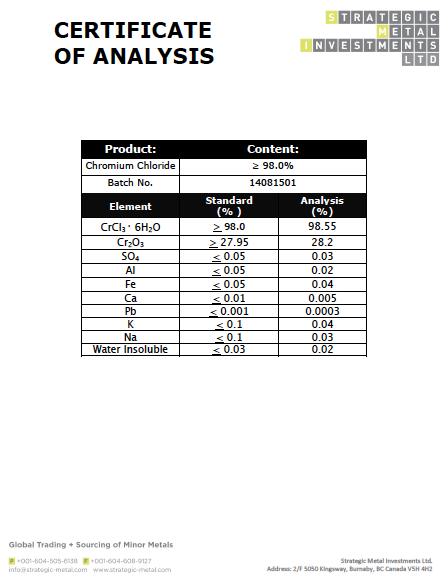 
The formula for an ionic compound must contain the same number of positive and negative charges so that the charges are balanced and it is neutral overall. Cations are positively charged ions, and anions are negatively charged ions. Chromium Chloride Molecular Formula CrCl3 Molecular Weight 158.36 g/mol Grams (g) HS Code 28273990 Boiling point 1,300 C Melting Point 1,152 C Density. The table shows the names and formulae of some common ions. There will be a chloride for each oxidation. The other fluoride of tin is SnF 4, which was previously called stannic fluoride but is now named tin(IV) fluoride.Ĭhemists use nomenclature rules to clearly name compounds.To deduce the formulae of ionic compounds, the formulae of their ions can be used. What is the chemical formula of Chromium Chloride hydrate Chromium has four oxidation states: 2, 3, 4, and 6. This represents the formula SnF 2, which is more properly named tin(II) fluoride. This is the chemical formula for chromium (III) nitrate: Cr (NO3) Calculate the mass percent of oxygen in chromium (III) nitrate. 
Round your answer to the nearest percentage. For example, you may see the words stannous fluoride on a tube of toothpaste. Science Chemistry This is the chemical formula for chromium (III) nitrate: Cr (NO3) Calculate the mass percent of oxygen in chromium (III) nitrate. Cr labeled sodium chromate or chromium chloride by gavage or intravenous injection and found that, regardless of the route, (51)Cr from chromium. Once again, notice that replacing water molecules by chloride ions changes the. A 0.025-g sample of a compound composed of boron and hydrogen, with a molecular mass of 28 amu, burns spontaneously when exposed to air, producing 0.063 g of B2O3. This happens when two of the water molecules are replaced by chloride ions to give the tetraaquadichlorochromium(III) ion - Cr(H 2 O) 4 Cl 2 . There will be a chloride for each oxidation state of. CrIII compounds do not cause chrome ulcerations but may cause allergic dermatitis in workers with prior sensitization to. Computed by PubChem 2.1 (PubChem release 2021.05.07) PubChem. In the presence of chloride ions (for example with chromium(III) chloride), the most commonly observed color is green. What is the chemical formula of Chromium Chloride hydrate Chromium has four oxidation states: 2, 3, 4, and 6. Though this naming convention has been largely abandoned by the scientific community, it remains in use by some segments of industry. ILO International Chemical Safety Cards (ICSC) CrNa 2 O 4. Out-of-date nomenclature used the suffixes – ic and – ous to designate metals with higher and lower charges, respectively: Iron(III) chloride, FeCl 3, was previously called ferric chloride, and iron(II) chloride, FeCl 2, was known as ferrous chloride. \): Names of Some Transition Metal Ionic Compounds Transition Metal Ionic Compound 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
